Is methanol soluble in organic solvents?
Then, is methanol an organic solvent?
Methanol, 2-propanol, and acetonitrile are widely used as organic solvents in MEKC.
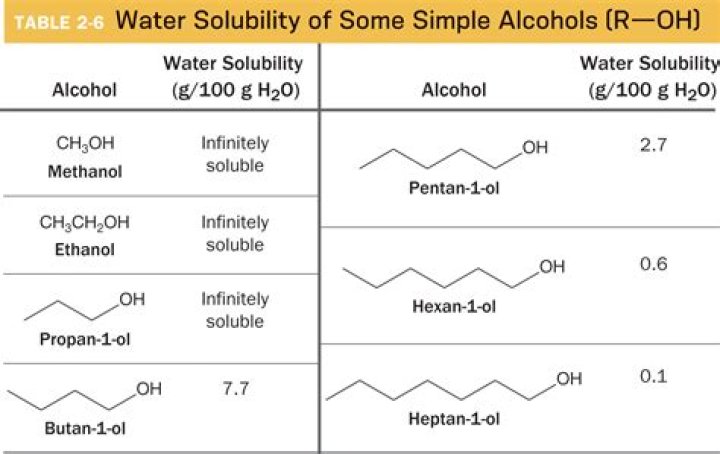
Also, what solvent can dissolve methanol? You find that the smaller alcohols - methanol, ethanol, and propanol - dissolve easily in water.
Keeping this in consideration, is methanol soluble or insoluble?
Methanol is soluble in water. Like water, methanol has relatively strong hydrogen bonding acting between its molecules. The attraction between methanol and water is strong enough to overcome the methanol-methanol and water-water intermolecular forces of attraction.
Is soluble in organic solvents?
Thus, most organic molecules are typically relatively non-polar and are usually soluble in organic solvents. (e.g. ethyl acetate, diethyl ether, dichloromethane, chloroform, petroleum ether, hexanes etc.) However, some organic molecules are more polar and therefore more soluble in water.
Related Question Answers
What is the formula of methanol?
CH3OHCan ethanol turn into methanol?
Methanol is formed in very small amounts during fermentation, the process by which alcohol is made from plant products like grape juice or cereal grains. Commercially-made spirits are very safe because manufacturers use technologies specifically designed to ensure methanol is separated from the ethanol.What are common organic solvents?
Examples of organic solvents- aromatic compounds, eg benzene and toluene.
- alcohols, eg methanol.
- esters and ethers.
- ketones, eg acetone.
- amines.
- nitrated and halogenated hydrocarbons.
What do organic solvents dissolve?
Organic solvents, such as chloroform/methanol mixtures are commonly used to dissolve membrane lipids and release the integral proteins and subcellular components. Organic solvent or mineral acid extraction does not, however, preserve morphological or metabolic integrity.What are natural solvents?
Organic solvents are carbon-based substances capable of dissolving or dispersing one or more other substances. Many classes of chemicals are used as organic solvents, including aliphatic hydrocarbons, aromatic hydrocarbons, amines, esters, ethers, ketones, and nitrated or chlorinated hydrocarbons.Is methanol a health hazard?
Methanol is highly flammable and toxic. Direct ingestion of more than 10mL can cause permanent blindness by destruction of the optic nerve, poisoning of the central nervous system, coma and possibly death. These hazards are also true if methanol vapors are inhaled.What is the pH of methanol?
In methanol, neutral is when H equals CH3O , which occurs when H is 10 8.3 or a pH of 8.3. Methanol–water mixtures have autoprotolysis constants between 14 (water) and 16.6 (methanol), so neutral in these mixtures ranges from pH 7 to pH 8.3.Is methanol a good solvent?
Methanol is a good solvent for extraction and it is frequently used in biology because of its polarity.Is water soluble in methanol?
To conclude, the strength of the attraction of the OH group makes methanol completely miscible in water. Any amount of methanol will usually dissolve in water. As for the limitation, the solubility of alcohols starts to decrease starting with the four-carbon butanol.Is salt soluble in methanol?
The attraction between the Na+ and Cl− ions in the solid is so strong that only highly polar solvents like water dissolve NaCl well.Aqueous solutions.
| Solubility of NaCl (g NaCl / 1 kg of solvent at 25 °C (77 °F)) | |
|---|---|
| Methanol | 14 |
| Ethanol | 0.65 |
| Dimethylformamide | 0.4 |
| 1-Propanol | 0.124 |
Is biphenyl soluble in methanol?
Biphenyl was only partially soluble in methyl alcohol because biphenyl has no polar functional groups like benzophenone does to make it slightly polar. Biphenyl is nonpolar while methyl alcohol has intermediate polarity, so only some of the biphenyl is able to dissolve in the methyl alcohol.Does KCL dissolve in methanol?
Potassium chloride (also known as Sylvite, KCl, or potassium salt) is a metal halide salt composed of potassium and chlorine.Solubility.
| Solvent | Solubility (g/kg of solvent at 25 °C) |
|---|---|
| H2O | 360 |
| Liquid ammonia | 0.4 |
| Liquid sulfur dioxide | 0.41 |
| Methanol | 5.3 |
Is salt soluble in kerosene?
I am Salt because salt dissolves in polar solvent i.e. water and is insoluble in non-polar solvent i.e. kerosene.Why ethanol is more soluble than methanol?
Ethanol has a 2 carbon chain and a OH group. Carbon chain on the other hand as nonpolar is repelled. Solubility of alcohols is therefore determined by the stronger of the two forces. Because of the strength of the attraction of the OH group, first three alcohols (methanol, ethanol and propanol) are completely miscible.Is LiCl soluble or insoluble?
In case of LiCl, the hydration energy is higher than the lattice energy. Hence, LiCl is water soluble. Due to higher polarization, LiCl has some covalent character. Hence, it is soluble in non polar solvents such as acetone.What is the solubility of methanol?
Methanol| Names | |
|---|---|
| Density | 0.792 g/cm3 |
| Melting point | −97.6 °C (−143.7 °F; 175.6 K) |
| Boiling point | 64.7 °C (148.5 °F; 337.8 K) |
| Solubility in water | miscible |
Is ethanol more polar than methanol?
My review book (Princeton review) says that ethanol has stronger intermolecular forces than methanol because it has a large molecular mass and is therefore more polarizable and more soluble in water than methanol. Therefore ethanol is less polar.What are the 10 examples of solvent?
Similarly sweet water has water as solvent and sugar as solute. Similarly some other examples of solute are salt, Lemon, oxygen, carbon dioxide, Nitrogen and ethyl alcohol. While examples of solvents are water, milk, Toluene, Acetone, ethanol, Glycerol, Petroleum,and Ether.Is Vinegar a solvent?
In vinegar, acetic acid is the solute and water is the solvent and in bleach, sodium hypochlorite is the solute and water is the solvent.What is the strongest solvent?
WaterIs Salt a solvent?
In a NaCl solution (salt-water), the solvent is water. A solute is the component in a solution in the lesser amount. In a NaCl solution, the salt is the solute. A solution may contain more than one solute.Does acetone dissolve sugar?
Water, for example, will dissolve salt or sugar. Some solvents, alcohol and acetone for example, are “miscible” with water meaning that they can be mixed with water in various proportions to create mixtures which may exhibit the solvent properties of both liquids.Does alcohol dissolve oil?
At ordinary temperatures even absolute alcohol is not a good solvent for vegetable oils since the solubility is even less than 10 g. of oil per 100 g. of alcohol. Mowrah, safflower, peanut, and cottonseed oils are soluble in absolute alcohol at 70°C.What are common solvents?
Common Solvents Used in Organic Chemistry: Table of Properties 1| Solvent | Formula | Solubility² |
|---|---|---|
| acetic acid | C2H4O2 | Miscible |
| acetone | C3H6O | Miscible |
| acetonitrile | C2H3N | Miscible |
| benzene | C6H6 | 0.18 |


